Separation Techniques
9/7
We started on a new topic today. It has a majestic name - Separation Techniques. Sure sounds like fun. Today I discovered about different types of separation techniques (though I still have not figured out how they work). I also learned about elements. The most interesting part of the experiment was the hands-on experiment on sulphur and iron fillings. I am still wondering about how the chemical reaction between sulphur and iron fillings occur. Today I experimented with sulphur and iron fillings. My hypothesis was that they will be chemically combined. I concluded that they became a compound after heating. My next experiment will be about crystalisation. Today I observed that crystals formed when the sulphur was cooled. I predict that crystals will be formed. I also measured how long it took for the crystals to be formed. I concluded that sulphur can form crystals.
Today I learned about crystalisation. I discovered that crystalisation is the formation of crystals. Today I observed crystalisation. I now know what happened to the sulphur after heating. I am still unsure about crystalisation. Today I conducted a science lab on sublimation. Another question that I have is, "How does sublimation take place?" Today we experimented on crystalisation. WeI observed that crystals formed when the sulphur was cooled. I predict that crystals will be formed. I also measured how long it took for the crystals to be formed. I concluded that sulphur can form crystals. Today I learned about crystalisation. I discovered that crystalisation is the formation of crystals. Today I observed crystallisation. Today I observed crystalisation. I now know what happened to the sulphur after heating. I am still unsure about crystallisation. Another question that I have is, "How does crystallisation work?"
Then we learnt how to light up the bunsen burner which was very very fun.
Today I learned about crystalisation. I discovered that crystalisation is the formation of crystals. Today I observed crystalisation. I now know what happened to the sulphur after heating. I am still unsure about crystalisation. Today I conducted a science lab on sublimation. Another question that I have is, "How does sublimation take place?" Today we experimented on crystalisation. WeI observed that crystals formed when the sulphur was cooled. I predict that crystals will be formed. I also measured how long it took for the crystals to be formed. I concluded that sulphur can form crystals. Today I learned about crystalisation. I discovered that crystalisation is the formation of crystals. Today I observed crystallisation. Today I observed crystalisation. I now know what happened to the sulphur after heating. I am still unsure about crystallisation. Another question that I have is, "How does crystallisation work?"
Then we learnt how to light up the bunsen burner which was very very fun.
10/7
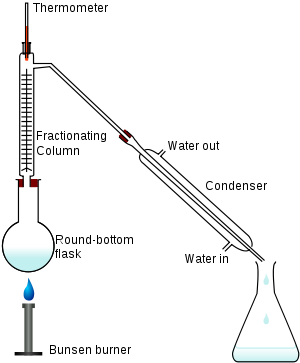
We went to the Chemistry Lab again today. Ms Goh showed us a simple distillation set-up. I was really really amazed at how it works. Look at the picture below. When I grow up, I want to research more about it as it looks very fun.
Anyway, moving on, Ms Goh then told us that there was more to the distillation technique. Other than, simple distillation there was also fractional distillation, which was separating a liquid from another liquid with a different boiling point. It is also in the picture below.
Anyway, moving on, Ms Goh then told us that there was more to the distillation technique. Other than, simple distillation there was also fractional distillation, which was separating a liquid from another liquid with a different boiling point. It is also in the picture below.
13/7
We stayed at home today for HBL. It was really fun. I think the school should have more HBL sessions as we not only could interact through the computer, it was also a more fun way of learning. I feel that the assignments were just the right time span for us to complete during the one hour block and my group completed it punctually. We planned an experimental procedure to separate Sodium Chloride, Magnesium Carbonate and Sulfur. I partnered with Natalie and Wei Xuan (my dear deskies) is as follows:
Group Members: Wei Xuan, Charissa Poh and Natalie Tan
Class 101, Group 2/B
Separation Techniques that we can consider using: (though not all can be used in this case)
- Filtration
- Crystallisation
- Simple Distillation
- Fractional Distillation
- Evaporation
- Bar Magnet
- Chromatography
The chemicals are Sodium Chloride, Magnesium Carbonate and Sulfur.
Notes (Relevant Information on the Chemicals): Sodium Chloride can dissolve in water. Sulfur sublimates and thus can be crystallised in a solution or on a surface. Magnesium carbonate is practically insoluble in water; dissolve the salt, filter the solution. Magnesium carbonate remain on the filter, the sodium chloride is now in solution. You can use this solution as table salt solution or by evaporation of the water you can obtain pure crystallized NaCl. However, it is simpler to buy pure sodium chloride; also, magnesium carbonate is not dangerous and is a common food additive.
Read more: http://wiki.answers.com/Q/How_do_you_remove_magnesium_carbonate_from_table_salt_to_make_the_salt_totally_pure#ixzz20SOlMCi0
Procedures
1. Make sure that the mixture is dry and place a funnel over the crucible.
2. Then, heat the mixture with a Bunsen Burner. The Sulfur will soon evaporate and it will crystallise on the surface of the funnel.
3. Afterwards, there are only sodium chloride and Magnesium Carbonate left in the crucible.
4. Pour water into the crucible and then pour the sodium chloride and magnesium carbonate solution into a beaker.
5. By then, the (salt) sodium chloride would have dissolved, except for the Magnesium Carbonate.
6. Lay a filter paper in another (not the one used to crystallise the sulfur) filter funnel and pour the solution in.
7. Afterwards, evaporate (you may consider using the Bunsen Burner to heat the solution but it is not compulsory) the water from the filtrate to obtain sodium chloride!
8. Then, we scrape off the sulfur crystals that are found on the sides of the funnel into another container. Also, collect the sodium chloride from the beaker and the magnesium carbonate from the filter paper.1. The mixture must be dry. The funnel to ensure that none of the sulfur will not escape during heating.
Explanation
1. The mixture must be dry. The funnel is needed to ensure that none of the sulfur will not escape during heating.
2. Sulfur can be crystallised, hence it can turn into vapour and condense as crystals when it is being heated.
3. Sulphur has the lowest boiling point, hence will evaporate faster and condense on the cooler surface of the funnel, forming crystals.
4. The water is poured into the crucible first to ensure that all the contents are removed from the crucible.
5. Sodium Chloride dissolves in water while Magnesium Carbonate does not.
6. The salt solution (sodium chloride) will be filtered out as the distillate while the Magnesium Carbonate is the residue which will not be filtered.
7. During evaporation, the water from the solution will evaporate due to the heat, leaving behind the sodium chloride.
8. Collect all the dry elements. These elements would be pure if the laboratory equipments are clean.
Group Members: Wei Xuan, Charissa Poh and Natalie Tan
Class 101, Group 2/B
Separation Techniques that we can consider using: (though not all can be used in this case)
- Filtration
- Crystallisation
- Simple Distillation
- Fractional Distillation
- Evaporation
- Bar Magnet
- Chromatography
The chemicals are Sodium Chloride, Magnesium Carbonate and Sulfur.
Notes (Relevant Information on the Chemicals): Sodium Chloride can dissolve in water. Sulfur sublimates and thus can be crystallised in a solution or on a surface. Magnesium carbonate is practically insoluble in water; dissolve the salt, filter the solution. Magnesium carbonate remain on the filter, the sodium chloride is now in solution. You can use this solution as table salt solution or by evaporation of the water you can obtain pure crystallized NaCl. However, it is simpler to buy pure sodium chloride; also, magnesium carbonate is not dangerous and is a common food additive.
Read more: http://wiki.answers.com/Q/How_do_you_remove_magnesium_carbonate_from_table_salt_to_make_the_salt_totally_pure#ixzz20SOlMCi0
Procedures
1. Make sure that the mixture is dry and place a funnel over the crucible.
2. Then, heat the mixture with a Bunsen Burner. The Sulfur will soon evaporate and it will crystallise on the surface of the funnel.
3. Afterwards, there are only sodium chloride and Magnesium Carbonate left in the crucible.
4. Pour water into the crucible and then pour the sodium chloride and magnesium carbonate solution into a beaker.
5. By then, the (salt) sodium chloride would have dissolved, except for the Magnesium Carbonate.
6. Lay a filter paper in another (not the one used to crystallise the sulfur) filter funnel and pour the solution in.
7. Afterwards, evaporate (you may consider using the Bunsen Burner to heat the solution but it is not compulsory) the water from the filtrate to obtain sodium chloride!
8. Then, we scrape off the sulfur crystals that are found on the sides of the funnel into another container. Also, collect the sodium chloride from the beaker and the magnesium carbonate from the filter paper.1. The mixture must be dry. The funnel to ensure that none of the sulfur will not escape during heating.
Explanation
1. The mixture must be dry. The funnel is needed to ensure that none of the sulfur will not escape during heating.
2. Sulfur can be crystallised, hence it can turn into vapour and condense as crystals when it is being heated.
3. Sulphur has the lowest boiling point, hence will evaporate faster and condense on the cooler surface of the funnel, forming crystals.
4. The water is poured into the crucible first to ensure that all the contents are removed from the crucible.
5. Sodium Chloride dissolves in water while Magnesium Carbonate does not.
6. The salt solution (sodium chloride) will be filtered out as the distillate while the Magnesium Carbonate is the residue which will not be filtered.
7. During evaporation, the water from the solution will evaporate due to the heat, leaving behind the sodium chloride.
8. Collect all the dry elements. These elements would be pure if the laboratory equipments are clean.
16/7
Today we did an experiment on chromatography. It was so fun. I was amazed by how the solvent could travel up the strip of paper without any help from humans. I was practically staring at it the whole time. The picture below shows the set-up of chromatography. It was so cool!
We also got back the crystals from last time. They were nice and shiny like stars in the night sky. We were all so mesmerised by the beauty of the of the shining jewels that we stretched our arms to grasp the magnificent jewelery but was stopped by Ms Goh. We looked up. She kindly reminded us not to touch the crystals. It is quite serious and dangerous if we touch the chemicals with our bare hands. Tragic.
We also got back the crystals from last time. They were nice and shiny like stars in the night sky. We were all so mesmerised by the beauty of the of the shining jewels that we stretched our arms to grasp the magnificent jewelery but was stopped by Ms Goh. We looked up. She kindly reminded us not to touch the crystals. It is quite serious and dangerous if we touch the chemicals with our bare hands. Tragic.
17/7
Today we stayed in class, sadly. I liked the lab a lot where we could do experiments. We went through the worksheets we had to complete in the pass few lab lessons. Then Ms Goh kindly invited two groups to present their HBL. They looked quite confident. Was really anticipating something.
But they did not really do it well as Miss Goh scolded them. Next lesson would be our turn to present. I sincerely hope she won't be angry with our group as we prepared for really long. 2 whole hours. Imagine what you could do in that 2 whole hours. I could eat at least 5 bowls of rice.
But they did not really do it well as Miss Goh scolded them. Next lesson would be our turn to present. I sincerely hope she won't be angry with our group as we prepared for really long. 2 whole hours. Imagine what you could do in that 2 whole hours. I could eat at least 5 bowls of rice.
19/7
Today was the last lesson of Separation Techniques. I am really really sad about it and I wish that it would prolong. Today many groups did the presentation except for 3 groups and we were one of them. We wanted to go but all our friends were so excited, elated and enthusiastic to go present so we did not get the chance. Anyway, as nice people we decided to let them go first although the thirst to go on stage was unbearable.
The next lesson will be on Monday, so as a very diligent person, I shall go revise the presentation thing again. I don't know about my group members, but I guess they will too, since they are so hardworking - almost as hardworking as me. Ahhh. I am nervous. I sincerely hope that we do not screw it up.
The next lesson will be on Monday, so as a very diligent person, I shall go revise the presentation thing again. I don't know about my group members, but I guess they will too, since they are so hardworking - almost as hardworking as me. Ahhh. I am nervous. I sincerely hope that we do not screw it up.
23/7
Today, sadly, we did not get to do the presentation again but we went to the lab! It was really fun and we did many experiments on elements and compounds and found out if they were chemical changes or physical changes. Once, my lab partner and I got into trouble for turning on the gas tap while walking off to get the lighter. Ms Goh gave us a piece of her mind and we were really sorry. We hung our heads low and continued with the other experiments.
I learnt an important lesson from today. No matter what we do in the lab, safety first. It is definitely the most important. Charlotte and I should not have left a flame unattended because someone might trip over something and fall towards the flame and SPLAT! It would be over.
I learnt an important lesson from today. No matter what we do in the lab, safety first. It is definitely the most important. Charlotte and I should not have left a flame unattended because someone might trip over something and fall towards the flame and SPLAT! It would be over.