Kinetic Particle Theory
1) The kinetic particle theory states that all matter is made up of tiny particles that are in constant, random motion.
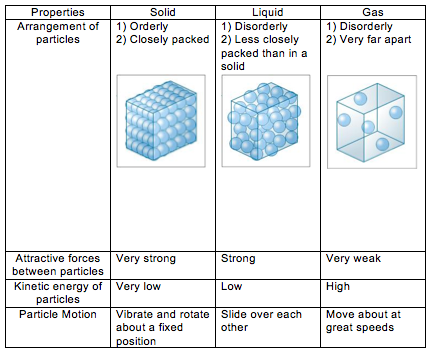
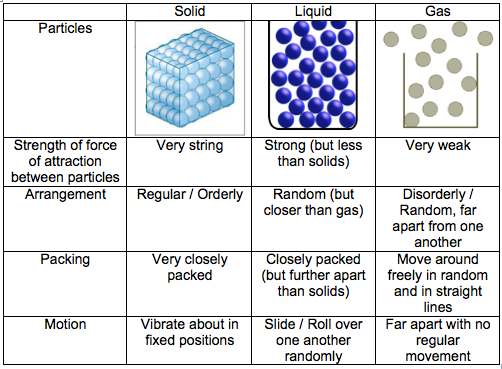
2) The differences in the states of matter can be explained in terms of the kinetic particle theory.
2) The differences in the states of matter can be explained in terms of the kinetic particle theory.
3) Particles in solids, liquids and gases have different amounts of kinetic energy. Gases have the highest energy content content, followed by liquids, and finally solids.
4) Changes of state occur when particles gain or lose energy.
4) Changes of state occur when particles gain or lose energy.
5) Gases with lower molecular masses diffuse faster than gases with higher molecular masses.
6) Diffusion is the movement of particles from a region of higher concentration to a lower concentration.
7) The relative rates of diffusion:
~ Solids: No diffusion
~ Liquids: Slow diffusion
~ Gases: Rapid diffusion
6) Diffusion is the movement of particles from a region of higher concentration to a lower concentration.
7) The relative rates of diffusion:
~ Solids: No diffusion
~ Liquids: Slow diffusion
~ Gases: Rapid diffusion
Kinetic Particle Theory
All matter is made up of tiny particles that are in constant random movement.
Assumptions
1) Particles in a gas are not attracted to one another.
2) Particles possess kinetic energy.
3) Higher temperature will result in a higher kinetic energy.
2) Particles possess kinetic energy.
3) Higher temperature will result in a higher kinetic energy.
Temperature
Temperature is the reflection of the average kinetic energy.
Characteristics
(Refer to Manual Journal for more details and pictures of the set-ups)